Our Aim
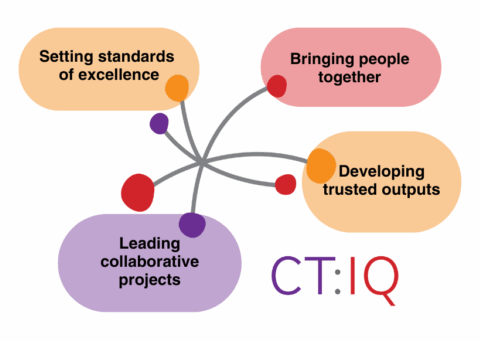
CT:IQ sets and maintains standards of excellence in clinical trials across Australia. CT:IQ’s diverse membership brings together all who are engaged in the Australian clinical trials endeavour, including industry, academia, government, health services, and consumers. Through a collaborative and multidisciplinary approach, we select and collaborate on projects that can positively impact research policy and practice across the sector. By design, our outputs are evidence-based, balanced, and trusted by decision-makers as best practice.
Year in Review
We are delighted to present CT:IQ's review of 2025, an exciting year where we have seen projects like the InFORMed template gain traction, and have released new outputs from projects like Beyond the Form and Flexible Trial Delivery. With a strong commitment to collaboration and a growing membership base, we are grateful for the support and participation of our members, both existing and new, and we look forward to another year of meaningful progress in 2026.