CT:IQ has now launched the outputs for this project, available here: Tools for Flexible Trial Delivery
BACKGROUND
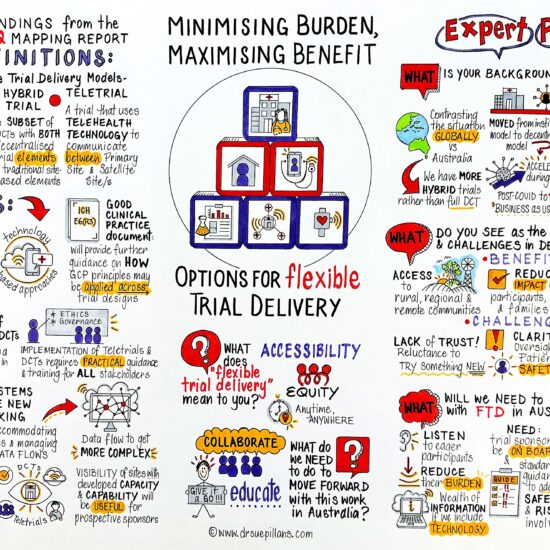
This project stemmed from a discussion about how to help sponsors and sites to increase the uptake of the great technologies and strategies that are now available to decrease participant burden in clinical trials. While Australia has had great success for strategies such as teletrials, there are other ways that aspects of trial activities can be adjusted to make them easier for a wider range of participants to take part.
The tools in this toolkit were developed by the Flexible Trial Delivery Team – a diverse group of professionals and consumers from across Australia. We also formed a participant sub-committee to specifically look at what participants need to get the most out of this way of working.
HEAR FROM OUR TEAM
Helen Monaghan
Naomi Strout
Gudrun Wells
WHAT WE DID

Toolkit released
Access Tools for Flexible Trial Delivery
Publications
Webinar
Watch our webinar on Flexible Trial Delivery, as experts explored the drivers, benefits, and challenges of flexible trial delivery options in Australia.
Artwork by Dr Sue Pillans